How to Calculate the Density of a Solid
Take a look at the two boxes below. Each box has the same volume. If each ball has the same mass, which box would weigh more? Why?
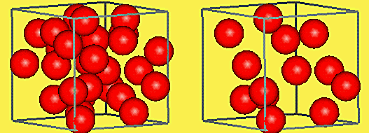
The box that has more balls has more mass per unit of volume. This property of matter is called density. The density of a material helps to distinguish it from other materials. Since mass is usually expressed in grams and volume in cubic centimeters, density is expressed in grams/cubic centimeter.
The box that has more balls has more mass per unit of volume. This property of matter is called density. The density of a material helps to distinguish it from other materials. Since mass is usually expressed in grams and volume in cubic centimeters, density is expressed in grams/cubic centimeter.
We can calculate density using the formula:
In the previous lesson you calculated the mass and volume of two blocks. If you would like to review these sections click on the appropriate links shown at the left.
Block I
Mass = 79.4 grams
Volume=29.8 cubic cm.
Block II:
Mass= 25.4 grams
Volume=29.8 cubic cm.
Now Let's calculate the density for each of these blocks.
Please enter your answer for Block I in the space provided:
Please enter your answer for Block II in the space provided:
What is each block made of?
Using the table below it is now possible for you to determine what substance makes up each block.The densities for some common substances are:
| Substance | Density (gm/cu.cm) |
| Air | 0.0013 |
| Wood (oak) | 0.85 |
| Water | 1.00 |
| Ice | 0.93 |
| Aluminum | 2.7 |
| Lead | 11.3 |
| Gold | 19.3 |
| Ethanol | 0.94 |
| Methanol | 0.79 |
Block I is made of:
Block II is made of :Go to Density of a Liquid Or search for more specific information on Mass -- Density -- Volume
- - -Or return to the Science and Math Activity Home Page
Mass Volume Density Activities
Mass: Learn how to measure the mass of an object using a triple beam balance
Mass vs. Weight: Mass and weight are often confused by many students. Learn the difference and try some challenging problems.
Volume: Measure volume using a graduated cylinder.
Density of a Solid: Learn to calculate the density of an unknown solid from knowing its mass and volume.
Density of a Liquid: Learn to calculate the density of an unknown liquid from knowing its mass and volume using a graduated cylinder and triple beam balance. Learn what a hydrometer is, and what it can do.
Density Challenge: Great page for gifted and talented students! Some excellent challenging problems.
Assessment: Twenty questions on mass, volume and density (two levels of difficulty). Your test is marked online.
Science Project Ideas: Ideas for science projects using mass, volume and density concepts learned from this module.
Mass Volume Density Lab Exercise: Problem: What is the relationship between water pressure and depth of water?
An Integrated Math Science and Art (STEAM) Activity- Mass, Volume Density Activity using the Gates Project from Central Park NYC.